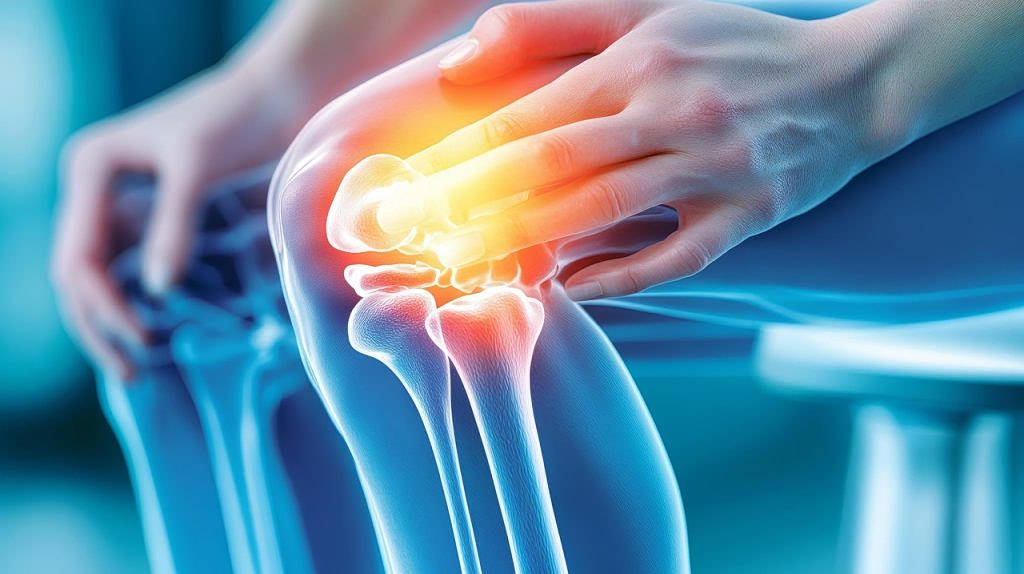
A research team in Thessaloniki has developed an innovative platform for personalized cartilage implants, marking a significant step toward new treatments for joint damage and degenerative conditions.
The technology, created at Aristotle University of Thessaloniki, is now entering the clinical trial phase after progressing through patent evaluation and regulatory approval procedures.
A new approach to tissue repair
The platform focuses on producing “living” implants tailored to each patient’s individual characteristics, including age, gender, body type and the nature of the injury.
Unlike traditional treatments that primarily manage symptoms, this method aims to restore damaged tissue itself. It is designed to address conditions such as traumatic injuries, arthritis and degeneration of spinal discs, which often lead to chronic pain and reduced mobility.
The product developed through this platform, known as NEOTICA, has already been classified as an advanced therapy medicinal product by National Organization for Medicines, the country’s regulatory authority for pharmaceuticals.
How the implants are created
The process begins with the collection of a small amount of fat tissue from the patient. From this, scientists isolate mesenchymal stem cells, which are then placed into a three-dimensional scaffold designed to replicate the damaged area.
Inside specialized laboratory conditions, these cells are guided to develop into cartilage-producing cells. Over time, they generate new tissue that can be implanted back into the body, where it integrates naturally.
Researchers describe the implants as “4D,” meaning they not only have a three-dimensional structure but also evolve over time. The scaffold gradually breaks down while the cells form new biological material, maintaining the strength and function of the tissue.
From laboratory to patients
So far, the technology has been tested in experimental models with promising results. The next step involves clinical studies in human patients suffering from chronic pain and degenerative conditions.
Depending on the severity of the damage, the implants could be applied through minimally invasive procedures or open surgery.
According to the research team, the necessary regulatory framework for clinical trials has already been secured, paving the way for the first applications in humans.